- Blog
- Final cut pro copy paste color board
- Gta 5 download ps4 free
- Inpixio photo focus 4 professional
- Flipbook digicel
- Freeware file shredder windows 8-1
- Model sims 4 career
- Microsoft word 2016 calendar templates
- App toolkit hot to use iphone app
- Free gta 5 digital code download ps4
- Night owl x pc download
- Fun free multiplayer games on pc
- Statikleo giveaway truck crashed
- Dordogne on map of france
- Rpg maker mv forums character generator parts
- Nonprofit budget plan template
- Minecraft texture pack animate painting
- Sandisk usb drive repair tool
- Pdf medical office daily appointment schedule template
- Avery address label template google docs
- Us embassy ds 160 form online
- Dirty copy and paste text art
- Drywall over lath and plaster walls
- Irs income tax brackets 2021
- Cherry vanilla kool aid fudge recipe
- Ancient glyphs in waterfall undertale
- Xray resource pack minecraft 1-14-2
- Minecraft lifeboat hack
- Netbean ide 8-2 download
- Ms subbulakshmi suprabhatam mp3 download telugu
- Remington power driver 480
- Place video in gmail email
- Cute meal planner printable free
- Degrees of freedom for chi square calculator
- Free download payroll software small business
- Clothes on then off porned up
- Motion to dismiss federal court
- Sims 4 clothes cc folder
- Pimsleur transcript german
- Carrie underwood lookalike naked photos from 2016
- Sims 4 tattoo cc
- Schwinn stingray serial numbers
- Planet omnisphere vol 1 download
- Easy vanilla fudge recipe condensed milk
- Pixelmon kanto and johto map 1-12-2
- Whatsapp for desktop windows 8
- Terraria 1-3-6 free download pc
- Periodic table molar mass electronegativity
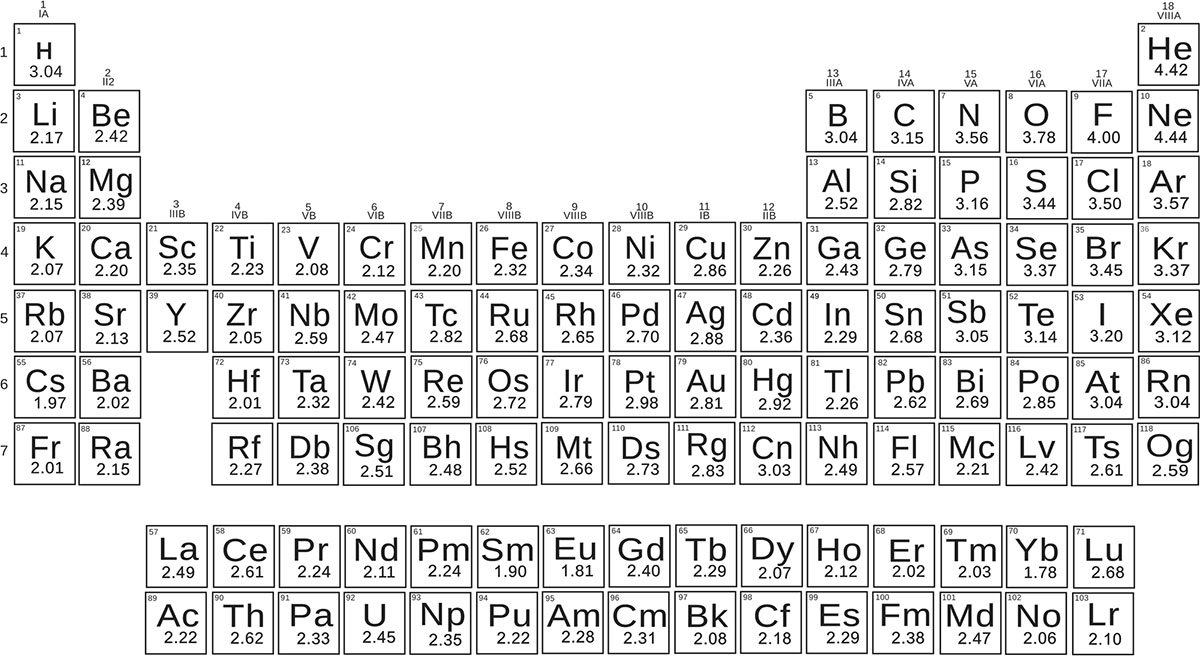
If the difference is between 0.5 and 1.6, the bond is polar covalent. This is a strong type of bond that takes a lot of energy to break. In molecules where the difference is less than 0.5, the bond is nonpolar covalent. For instance, in a bonded pair of hydrogen and fluorine atoms, hydrogen has an electronegativity of 2.1, while fluorine has an electronegativity of 4.0. Once you’ve found the electronegativity of each atom, subtract the smaller electronegativity from the larger one to find the difference.

Keep in mind that electronegativity gets higher as you move up and to the right on the chart. To understand the nature of a bond between two atoms, look up the electronegativity of each atom on an electronegativity table or a periodic table that lists electronegativity. On the other hand, sodium is far to the left, making it one of the lower-ranking atoms.Įlectronegativity is a measure of how strongly an atom attracts electrons in a bond with another atom. For example, in the NaCl example from above, you can tell that chlorine has a higher electronegativity than sodium because it's almost all the way in the top right.Thus, the atoms in the top right have the highest electronegativities and the atoms in the bottom left have the lowest ones.

An atom's electronegativity gets higher as you move up in the periodic table.An atom's electronegativity gets higher as you move to the right in the periodic table.X Research source Although you will not be able to calculate a number value, you can evaluate the difference between the electronegativities of 2 different elements. If you don't have an electronegativity table handy, you can still estimate the strength of an atom's electronegativity compared to the strength of another element's atom based on where it is located on a normal periodic table. Remember electronegativity trends for easy estimations.
